Findings From the REVEAL Registry*
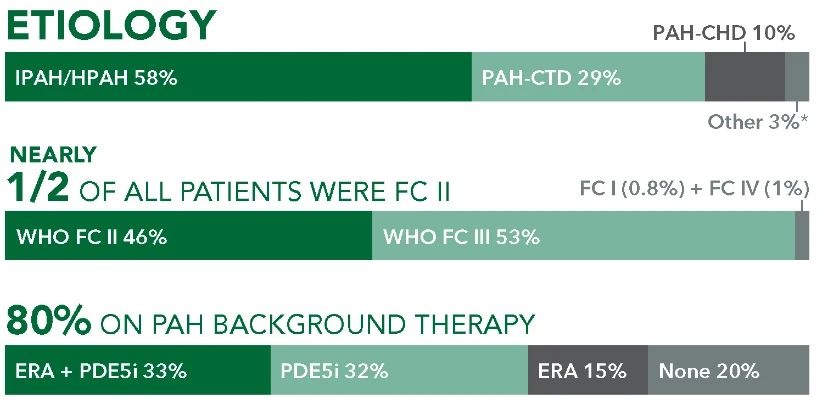
REVEAL was a US-based, observational registry involving 55 academic- and community-based treatment centers. 3515 patients enrolled between March 2006 and December 2009.4


REVEAL was a US-based, observational registry involving 55 academic- and community-based treatment centers. 3515 patients enrolled between March 2006 and December 2009.4





Clicking CONTINUE below will take you to the selected site, the content for which Johnson & Johnson is not responsible and to which this Privacy Policy does not apply. We encourage you to read the Privacy Policy of every online service you visit.



The following form is intended for use with patients who are eligible for VA benefits only.



This article includes information that has not been approved by the Food and Drug Administration for UPTRAVI® (selexipag). Please see full Prescribing Information available on this website. Authors[s] of this article have received remuneration from Actelion Pharmaceuticals US, Inc., or its affiliates.