


You are now leaving UptraviHCP.com
Clicking CONTINUE below will take you to the selected site, the content for which Johnson & Johnson is not responsible and to which this Privacy Policy does not apply. We encourage you to read the Privacy Policy of every online service you visit.



Confirm that you wish to download the Veterans Affairs (VA) Patient Enrollment Form for UPTRAVI® (selexipag)
The following form is intended for use with patients who are eligible for VA benefits only.

GRIPHON Is the Largest PAH Pivotal Trial With the Longest Endpoint Evaluation Period of Any Prostacyclin Pathway Therapy1-3
- Multicenter, long-term, double-blind, placebo-controlled, parallel-group, event-driven, phase 3 trial1,4
Study Design1-3


1156 PATIENTS Largest PAH pivotal trial UPTRAVI" (selexipag): n-574 | Placebo: n-582
1.4 YEARS (median duration of UPTRAVI*) Longest endpoint evaluation period of any prostacyclin pathway therapy
Baseline Patient Characteristics1,5
- Mean age: 48 years
- Female: 80%
- Median time from PAH diagnosis in patients taking UPTRAVI®: 0.9 years
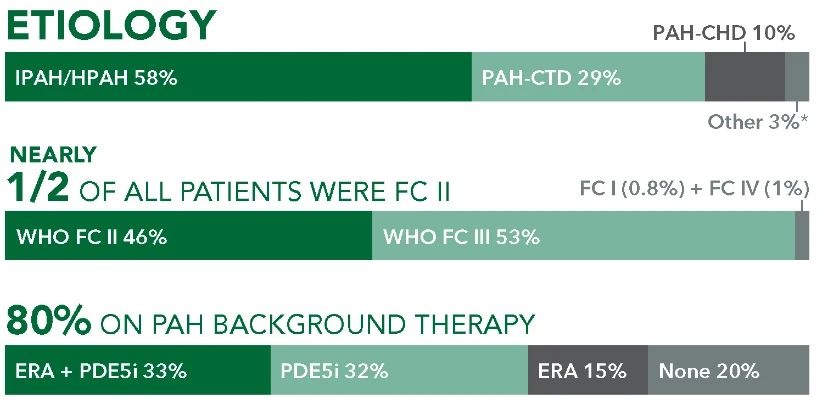
ETIOLOGY
IPAH/HPAH 58%
PAH-CHD 10%
PAH-CTD 29%
Other 3%*
NEARLY 1/2 OF ALL PATIENTS WERE FC II
WHO FC II 46%
FC I (0.8%) + FC IV (1%)
WHO FC III 53%
80% ON PAH BACKGROUND THERAPY
ERA + PDE5i33%
PDE5i 32%
ERA 15%
None 20%
This article includes information that has not been approved by the Food and Drug Administration for UPTRAVI® (selexipag). Please see full Prescribing Information available on this website. Authors[s] of this article have received remuneration from Actelion Pharmaceuticals US, Inc., or its affiliates.
