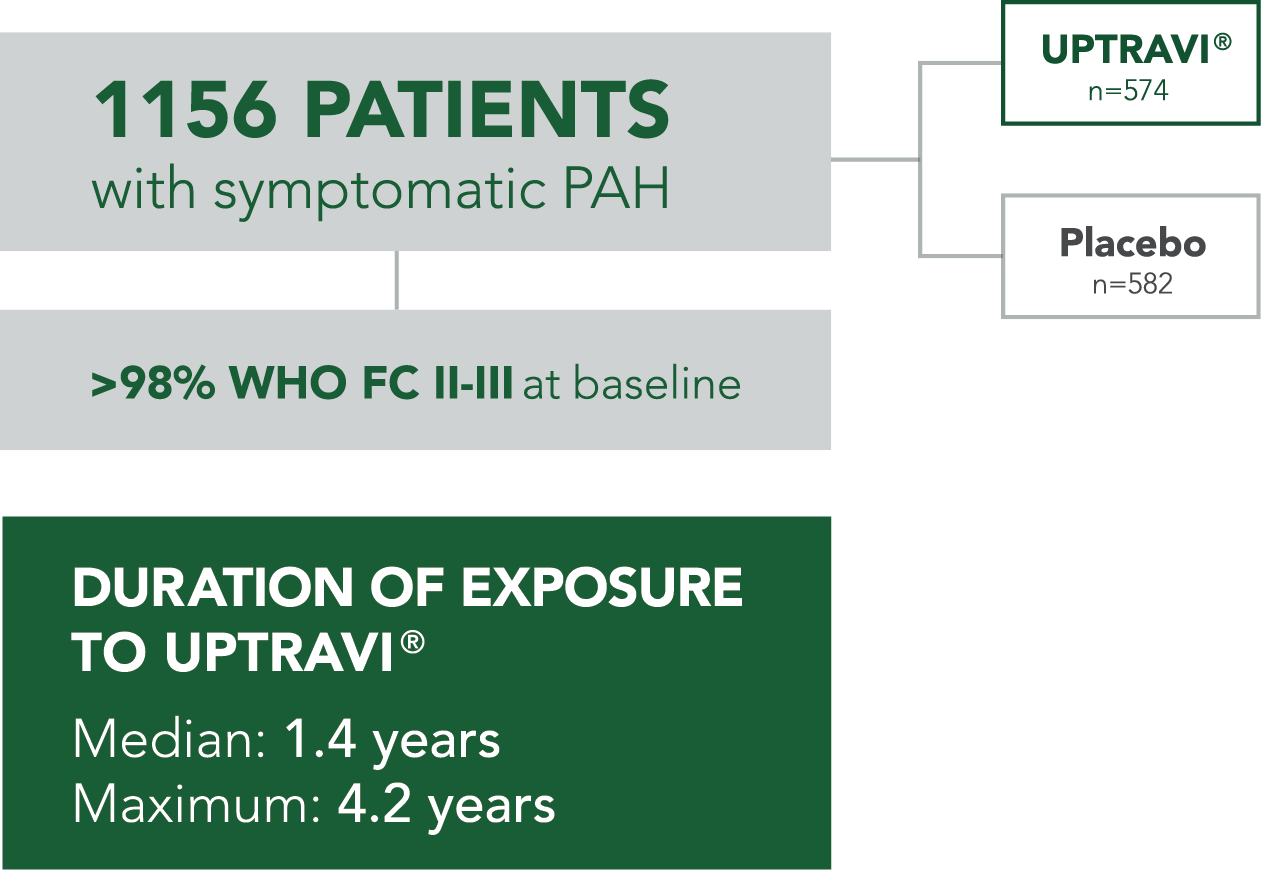
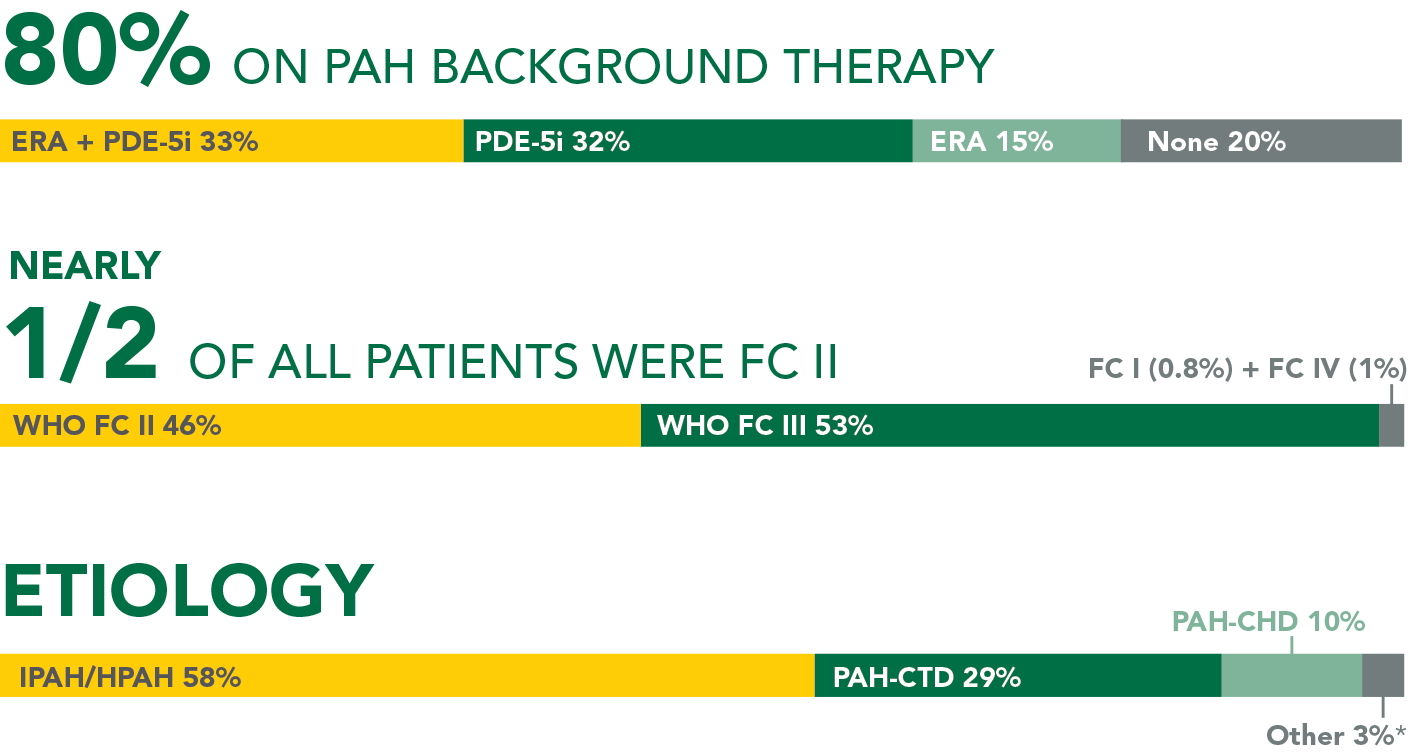
ADVERSE REACTIONS OCCURRING MORE FREQUENTLY WITH UPTRAVI® COMPARED WITH PLACEBO BY ≥3% IN THE GRIPHON TRIAL
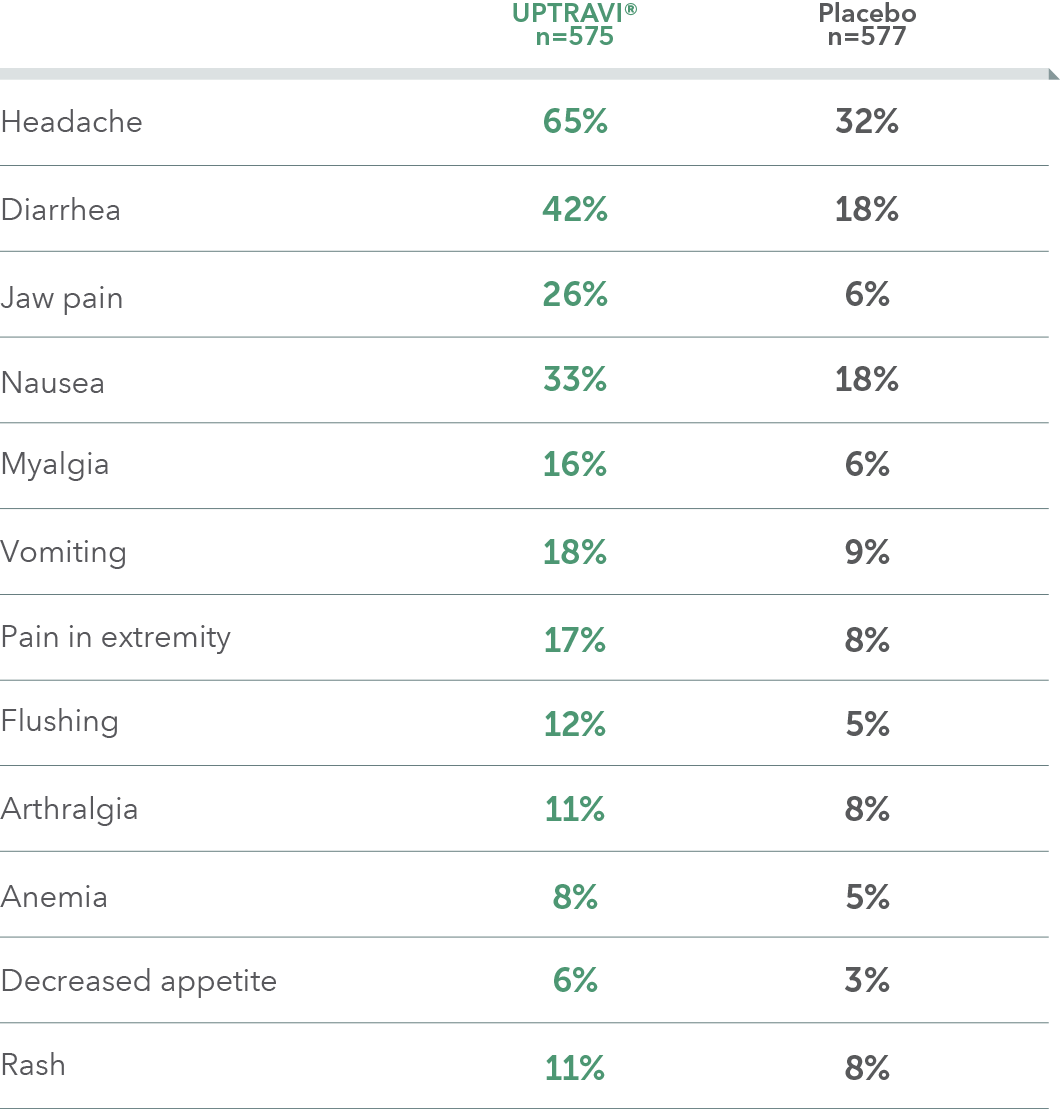
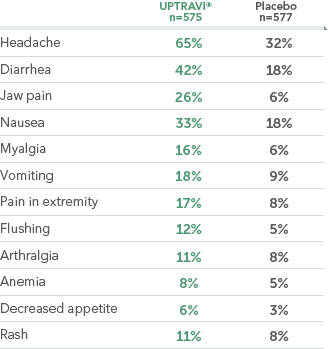
- These adverse reactions were more frequent during the titration phase
- Hyperthyroidism was observed in 1% (n=8) of patients on UPTRAVI® and in none of the patients on placebo
- Median duration of exposure to UPTRAVI® was 1.4 years
Postmarketing Experience
The following adverse reactions have been identified during postapproval use of UPTRAVI®.
Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
- Vascular disorders: symptomatic hypotension
Help your patients be prepared for potential adverse reactions and how to manage them
- Adverse Reactions Management Tool: Learn about adverse reactions common to prostacyclin-class therapies
- UPTRAVI® Dose Adjustment Phase Guide for patients: Use this guide for patients who are starting UPTRAVI® to help set goals and expectations for treatment. After discussion, give patients the guide to help them track and share how they’re feeling